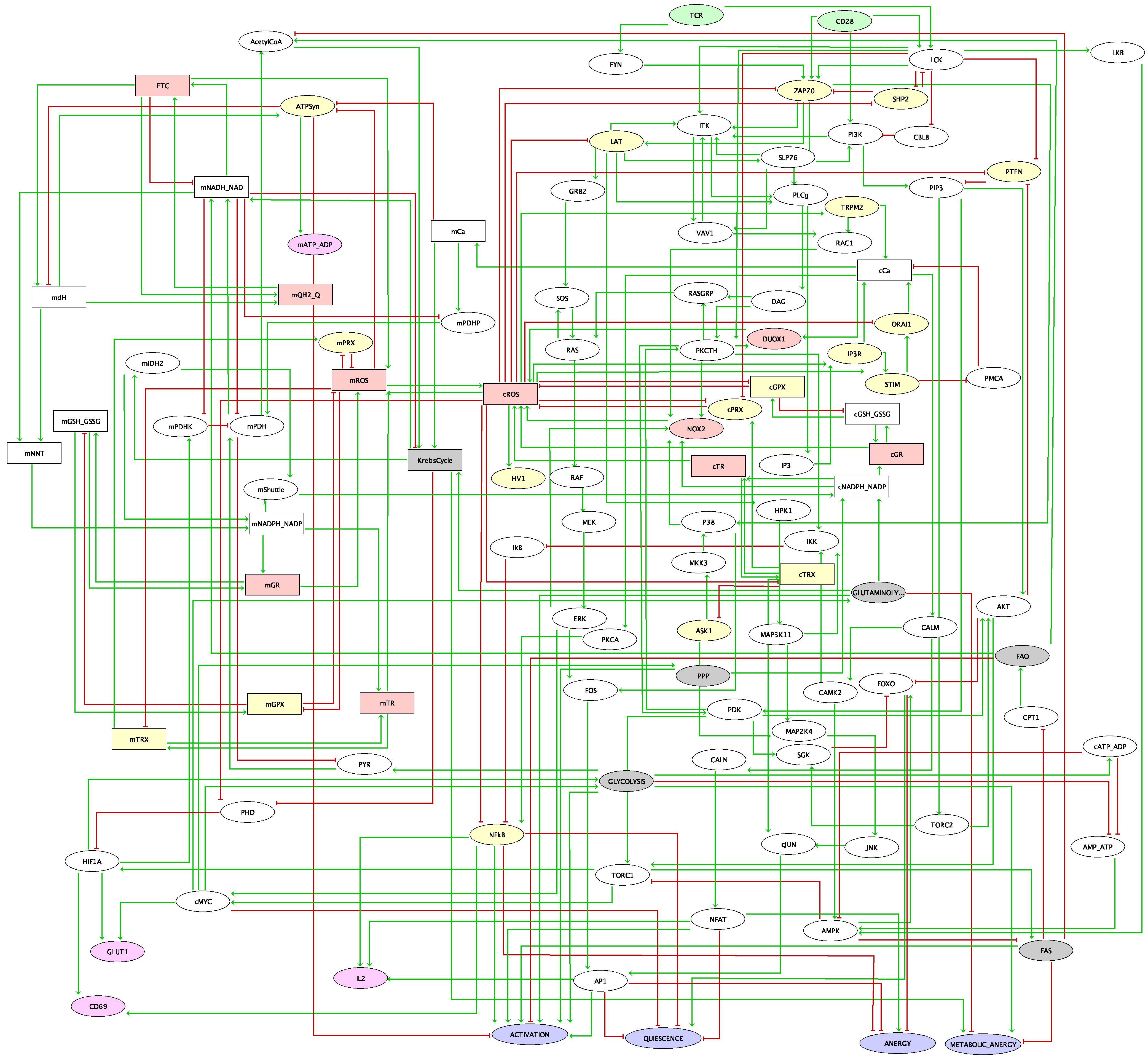
After the success of the new generation of immune therapies, immune checkpoint receptors have become one important center of attention of molecular oncologists. The initial success and hopes of anti-programmed cell death protein 1 (anti-PD1) and anti-cytotoxic T-lymphocyte-associated protein 4 (anti-CTLA4) therapies have shown some limitations since a majority of patients have continued to show resistance. Other immune checkpoints have raised some interest and are under investigation, such as T cell immunoglobulin and ITIM (immunoreceptor tyrosine-based inhibition motif) domain (TIGIT), inducible T-cell costimulator (ICOS), and T cell immunoglobulin and mucin domain-containing protein 3 (TIM3), which appear as promising targets for immunotherapy. To explore their role and study possible synergetic effects of these different checkpoints, we have built a model of T cell receptor (TCR) regulation including not only PD1 and CTLA4, but also other well studied checkpoints (TIGIT, TIM3, lymphocyte activation gene 3 (LAG3), cluster of differentiation 226 (CD226), ICOS, and tumour necrosis factor receptors (TNFRs)) and simulated different aspects of T cell biology. Our model shows good correspondence with observations from available experimental studies of anti-PD1 and anti-CTLA4 therapies and suggest efficient combinations of immune checkpoint inhibitors (ICI). Among the possible candidates, TIGIT appears to be the most promising drug target in our model. The model predicts that signal transducer and activator of transcription 1 (STAT1)/STAT4-dependent pathways, activated by cytokines such as interleukin 12 (IL12) and interferon gamma (IFNG), could improve the effect of ICI therapy via upregulation of Tbet, suggesting that the effect of the cytokines related to STAT3/STAT1 activity is dependent on the balance between STAT1 and STAT3 downstream signalling.